 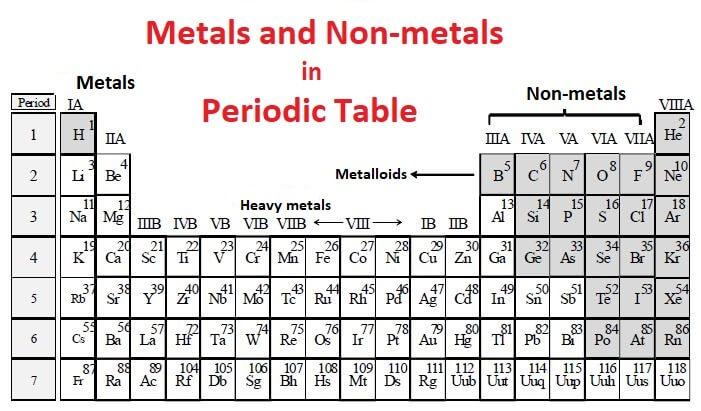
Note that there is no clear division between metallic and non-metallic character. Solution Non-metal in the periodic table: The non-metals are the elements that are located on the right side of the periodic table. Nonmetals in Biology AN IDIOSYNCRATIC VIEW OF THE PERIODIC TABLE. On descending a group in the periodic table, on average the outermost electrons get further and further away from the nucleus and as a result, become easier to remove from the atom. Fluorine gas reacts explosively with many other elements and compounds, and is considered to be one of the most dangerous known substances. most abundant transition metal in the ocean and is certainly beneficial and probably. Whats more, to the surprise of most chemists and going against the well-known trends of the periodic table, it turns out that francium is not the most reactive metal. It is not found in nature as a free element. The further down the group you go, the less reactive the element is.  
The most reactive element from group seven is fluorine which is at the top of that section of the periodic table. Since the noble gases are a special group because of their lack of reactivity, the element fluorine is the most reactive nonmetal. The halogens include fluorine, chlorine, bromine, iodine and astatine. The most reactive nonmetals reside in the upper right portion of the periodic table. Nonmetals tend to gain electrons in chemical reactions, and have a high attraction for electrons within a compound. Despite being a non-metal, hydrogen is often included in the reactivity series since it helps compare the reactivities of the metals. In addition, the atomic radius increases going down a group, placing the outer electrons further away from the nucleus and making that electron less attracted by the nucleus. There are many divisions in the periodic table, but one of the most important is between the metals close metal Shiny element that is a good conductor of electricity and heat, and which forms. Since the ionization energy decreases going down a group (or increases going up a group), the increased ability for metals lower in a group to lose electrons makes them more reactive. The metallic character increases as you go down a group. Reactivity of metals is based on processes such as the formation of halide compounds with halogens, and how easily the element displaces hydrogen from dilute acids. (Credit: User:Mirek2/Wikimedia Commons Source: Commons Wikimedia, Periodic Trends(opens in new window) License: Public Domain) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
